44 worksheet electrons in atoms
Gumdrop Atoms - Activity - TeachEngineering Feb 25, 2020 · Students use gumdrops and toothpicks to make lithium atom models. Using these models, they investigate the makeup of atoms, including their relative size. Students are then asked to form molecules out of atoms, much in the same way they constructed atoms out of the particles that atoms are made of. Students also practice adding and subtracting electrons from an atom and determining the overall ... Lewis Dot Structure – Easy Hard Science - learnwithdrscott.com The NH 3 Lewis structure has the typical case of nitrogen N in the center with 3 bonds to 3 other atoms. There is also a lone pair of electrons (unbonded electrons) on N. The lone pair (dots on N) could be drawn in any position… up, down, left, or right. Normally, however, we draw N with a pair of dots on top.
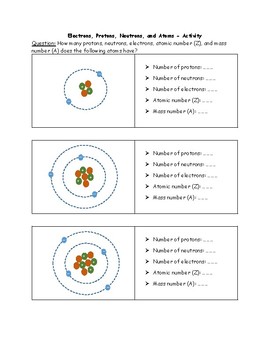
Isotope Worksheet Answer Key - ISD 622 # of electrons mass # a35 33 # of protons 19 # of neutrons 22 Isoto e name uranium-235 uranium-23 8 boron- 10 boron-11 atomic # Phos hocus-33 15 Write the hyphen notation and the nuclide (nuclear) symbol for an isotope that has 17 protons, 17 electrons, and 20 neutrons. 37 Isotopes are atoms of the same element with a different number of

Worksheet electrons in atoms
Molecule Shapes - VSEPR | Lone Pairs | Bonds - PhET ... Explore molecule shapes by building molecules in 3D! How does molecule shape change with different numbers of bonds and electron pairs? Find out by adding single, double or triple bonds and lone pairs to the central atom. Then, compare the model to real molecules! Loading... - BrainPop Loading... - BrainPop ... Loading... electron | Definition, Mass, & Facts | Britannica 22.09.2022 · electron, lightest stable subatomic particle known. It carries a negative charge of 1.602176634 × 10−19 coulomb, which is considered the basic unit of electric charge. The rest mass of the electron is 9.1093837015 × 10−31 kg, which is only 11,836the mass of a proton. An electron is therefore considered nearly massless in comparison with a proton or a neutron, and …
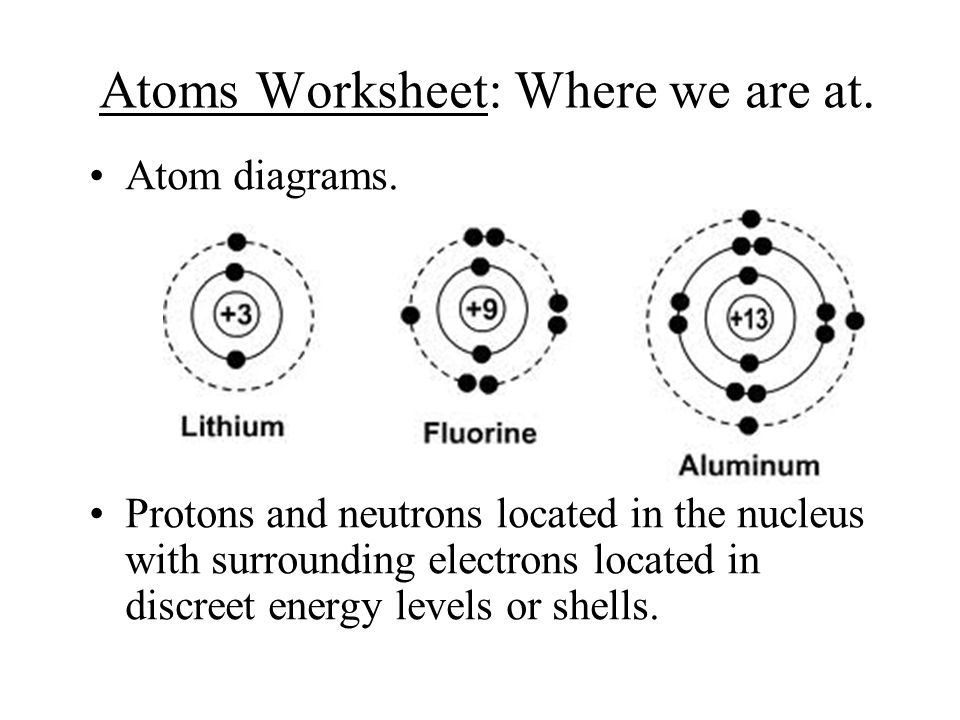
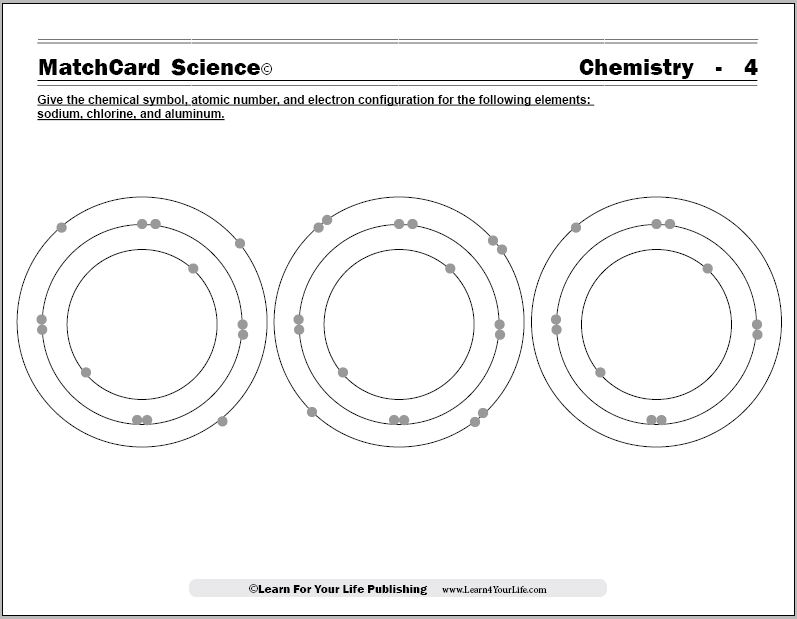
Worksheet electrons in atoms. Clickable Periodic Table of the Elements - ThoughtCo Feb 28, 2020 · Groups . Most periodic tables are color-coded so that you can see at a glance which elements share common properties with each other. Sometimes these clusters of elements (e.g., alkali metals, transition metals, non-metals) are called element groups, yet you'll also hear chemists refer to the columns (moving top to bottom) of the periodic table called element groups. Covalent Bonds vs Ionic Bonds - Difference and Comparison | Diffen For example, let us consider a Methane molecule i.e.CH 4. Carbon has 6 electrons and its electronic configuration is 1s22s22p2, i.e. it has 4 electrons in its outer orbit. According to the Octate rule ( It states that atoms tend to gain, lose, or share electrons so that each atom has full outermost energy level which is typically 8 electrons.), to be in a stable state, it needs 4 more … Oxygen - Element information, properties and uses | Periodic ... Today one gram can be beaten into a square meter sheet just 230 atoms thick, one cubic centimetre would make a sheet 18 square meters, 1 gram could be drawn out to make 165 meters of wire just 1/200 th of a millimetre thick. The gold colour in Buckingham Palace fence is actually gold; gold covered because it lasts 30 years; whereas gold paint ... Chemistry of Matter - Science Spot 12. How many electrons can each level hold? 1st = 2 2nd = 8 3rd = 18 13. What term is used for the electrons in the outermost shell or energy level? VALENCE 14. Scientists use two types of diagrams to show the electron configuration for atoms. Follow your teacher’s directions to complete the diagrams. Sulfur Atomic # = 16 Atomic Mass = 32 ...
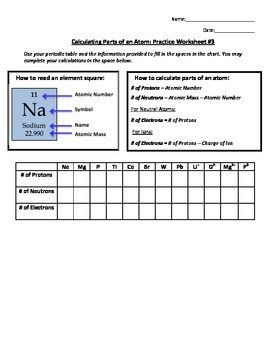
Atom Worksheets Atom is the most basic unit of matter. It has a dense nucleus with a cloud of negatively charged electrons surrounding it. Here are the parts of an Atom: Electron; it is a subatomic particle with a negative electrical charge. The mass of an electron is so small that it is generally not even considered. An Atom Apart see atoms with microscopes, they have developed ways to detect them and learn about them. Atoms are made up of three basic parts; protons, neutrons, and electrons. There is a core, or nucleus, and an electron cloud. The nucleus is made up of positively charged protons and neutral neutrons. The nucleus is held closely together by electromagnetic ... Basic Atomic Structure Worksheet Key - Neshaminy School District Give the symbol of and the number of electrons in a neutral atom of: Uranium Boron Chlorine Iodine Xenon Give the symbol of and the number of neutrons in one atom of: (Mass numbers are ALWAYS whole numbers...show your calculations) Barium Carbon Fluorine Europium IS-I 10 Bismuth Hydrogen Magnesium Mercury electron | Definition, Mass, & Facts | Britannica 22.09.2022 · electron, lightest stable subatomic particle known. It carries a negative charge of 1.602176634 × 10−19 coulomb, which is considered the basic unit of electric charge. The rest mass of the electron is 9.1093837015 × 10−31 kg, which is only 11,836the mass of a proton. An electron is therefore considered nearly massless in comparison with a proton or a neutron, and …
Loading... - BrainPop Loading... - BrainPop ... Loading... Molecule Shapes - VSEPR | Lone Pairs | Bonds - PhET ... Explore molecule shapes by building molecules in 3D! How does molecule shape change with different numbers of bonds and electron pairs? Find out by adding single, double or triple bonds and lone pairs to the central atom. Then, compare the model to real molecules!
































0 Response to "44 worksheet electrons in atoms"
Post a Comment