38 redox reaction worksheet with answers
25 Redox Review Worksheet Answers Scientific Notation Word Problems Redox Reactions Word Problem Worksheets . Pin By Quinna On Delete Igka Homan In 2021 Chemistry Redox Reactions Oxidation State . Half Reaction Of Wo2 To Wo3 Please Explain Yahoo Answers Yahoo Answers Answers Reactions .
For each reaction below, identify the atom oxidized, the atom reduced, the oxidizing agent, and the reducing agent. 1) Mg + 2HCl ( MgCl2 + H2. 2) 2Fe + 3V2O3 ( Fe2O3 + 6VO. 3) 2KMnO4 + 5KNO2 + 3H2SO4 ( 2MnSO4 + 3H2O + 5KNO3 + K2SO4. ... Oxidation Reduction Worksheet Answers ...
Redox Reactions Answer Key Redox Reactions CHEM 10 Review Worksheet The questions on this worksheet are both Chem 10 and Chem 11 level questions. The reaction that takes place in a chemical cell is best classi ed as A. Redox Reactions Worksheet Pdf Printable worksheets are a valuable lecture room tool. Oxidizedreducing agent O0 to O2-.

Redox reaction worksheet with answers
Practice Problems: Redox Reactions. Determine the oxidation number of the elements in each of the following compounds: a. H 2 CO 3 b. N 2 c. Zn(OH) 4 2-d. NO 2-e. LiH f. Fe 3 O 4 Hint; Identify the species being oxidized and reduced in each of the following reactions: a. Cr + + Sn 4+ Cr 3+ + Sn 2+ b. 3 Hg 2+ + 2 Fe (s) 3 Hg 2 + 2 Fe 3+ c. 2 As ...
Worksheet 25 - Oxidation/Reduction Reactions Oxidation number rules: Elements have an oxidation number of 0 Group I and II - In addition to the elemental oxidation state of 0, Group I has an oxidation state of +1 and Group II has an oxidation state of +2. Hydrogen -usually +1, except when bonded to Group I or Group II, when it forms hydrides, -1. ...
Balancing Redox Reactions Worksheet 1 Balance each redox reaction in . acid. solution. Mn 2+ + BiO3 -Æ MnO4 -+ Bi 3+ MnO4 -+ S2O3 2- Æ S4O6 2- + Mn 2+
Redox reaction worksheet with answers.
Chapter 20 Worksheet: Redox I. Determine what is oxidized and what is reduced in each reaction. Identify the oxidizing agent and the reducing agent, also. 1. 2Sr + O2 2SrO 2. 2Li + S Li2S 3. 2Cs + Br2 2CsBr 4. 3Mg + N2 Mg3N2 5. 4Fe + 3O2 2Fe2O3 6. Cl2 + 2NaBr 2NaCl + Br2 7. Si + 2F2 SiF4 8. 2Ca + O2 2CaO 9.
Oxidation Reduction Worksheet. Determine the oxidation number of each atom in the following substances. NF3 N +3 F -1 K2CO3 K +1 C 4 O -2 c. NO3- N____+5_____ O____-2_____ HIO4 H +1 I +7 O -2 For the following balanced redox reaction answer the following questions. 2 Fe+2(aq) + H2O2(aq) ( 2Fe+3(aq) + 2 OH-1(aq) What is the oxidation state of ...
Redox reactions practice problems determining oxidation numbers worksheet answers. For example the oxidation number of the oxygen in the oxide ion o 2 is 2. 1 This problem poses interesting problems especially with the Cl. Fe HCl —. In which substance is the oxidation number of. The oxidation number of hydrogen in a compound is 1 4.
Worksheet 1 - Oxidation/Reduction Reactions Oxidation number rules: Elements have an oxidation number of 0 Group I and II - In addition to the elemental oxidation state of 0, Group I has an oxidation state of +1 and Group II has an oxidation state of +2. Hydrogen -usually +1, except when bonded to Group I or Group II, when it forms hydrides, -1. ...
3. Balance the spontaneous redox reaction below. A spontaneous reaction is a reaction that occurs: 1) by a driving force that favors the product, 2) the free energy of the product is lower than the free energy of the reactant, and/or 3) occurs without any outside 'help' such as electrolysis. Identify the entities reduced and oxidized.
Balancing redox reactions worksheet 1 answer key the following redox reaction takes place in acidic solution. You can in a half reaction but remember half reactions do not occur alone they occur in reduction oxidation pairs 2 here are the correct half reactions. Mn 2 bio3 æ mno4 bi 3 mno4 s2o3 2 æ s4o6 2 mn 2 clo3 cl æ cl2 clo2.
Balancing redox reactions worksheet 1 balance each redox reaction in. Fe 2 o 3 3 co 2 fe 3 co 2 in acidic solution c. H 2o 2 cr 2o 7 2 o 2 cr 3 9. A trick to get around this is to balance any troublesome half reaction or the entire redox reaction first as if it were in acid using h and h 2 o.
Balancing Redox Equations Method 2: Half-reaction method 1. Divide the skeleton reaction into two half-reactions, each of which contains the oxidized and reduced forms of one of the species 2. Balance the atoms and charges in each half-reaction - Atoms are balanced in order: atoms other than O and H, then O, then H
Balancing redox reactions in basic solution. If the redox reaction was carried out in basic solution (i.e. alkaline conditions), then we have to put in an extra step to balance the equation. The steps for balancing redox reactions in basic solution are: Identify the pair of elements undergoing oxidation and reduction by checking oxidation states
Recognizing redox reactions worksheet answers. 3mg n2 mg3n2 5. Balance each of the following half cell reactions. Suited for student in y10 and y11. In the reaction mg cl2 mgcl2 the correct half reaction for the oxidation that occurs is a. Redox reactions are a chemical reaction in which electrons are exchanged through oxidation and reduction.
24. In the reaction Mg+Cl2!MgCl2, the correct half-reaction for the oxidation that occurs is A. Mg+2e !Mg2+ B. Cl2 +2e !2Cl C. Mg !Mg2+ +2e D. Cl2!2Cl +2e 25. The reaction that takes place in a chemical cell is best classi ed as A. fusion B. redox C. transmutation D. cracking 26. Which equation represents the half-reaction that takes place at ...
Questions 12-13: Balancing redox reaction equations is a skill which combines chemical knowledge, common sense, and intuition. There are many methods available for balancing redox reactions, A number have been brought to your attention including the one that follows. Choose a method and complete questions 12-13.
Balancing redox reactions worksheet with answers pdf. Zn no 3 zn2 nh4 3. In the first case you separate out the oxidation and reduction half reaction and in the second case you do it all at once. Be sure the reaction is redox look at the oxidation numbers for the atoms in the reaction. Teo 3 2 n 2o 4 te no 3 10.
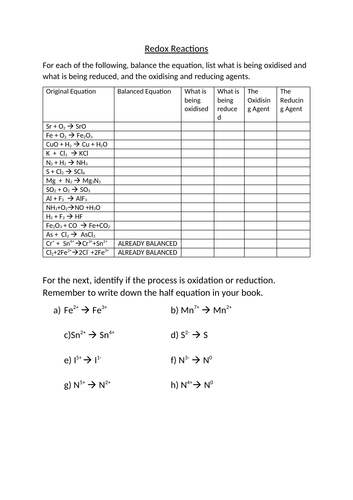
2-5 Redox Reactions Practice Worksheet With Answers - Free download as PDF File (.pdf), Text File (.txt) or read online for free. useful for igcse chapter redox reactions
reaction and reduction half-reaction, identify the oxidizing and reducing agents and any spectator ions. Half - Reactions Homework ... Balance the following redox reactions by the half-reaction method, rewriting the balanced equations below the given unbalanced equation. Show your work below each reaction and
Mno 2 mn 2o 3 balance each redox reaction in acid solution using the half reaction method. Redox reactions worksheet answers. A redox reaction always involves a. Oxidation is associated with electron loss helpful mnemonic. H 2o 2 cr 2o 7 2 o 2 cr 3 9. Determination of activity of some metals by reaction with hydrogen ion doc 28 kb redox ...
Redox Reactions Worksheet Answer Key. Mg0 2H1 Cl-1 Mg1 Cl2-1 H20 Mg is oxidized RA. Determination of activity of some metals by reaction with hydrogen ion doc 28 kb redox. In the reaction mg cl2 mgcl2 the correct half reaction for the. H 2o 2 cr 2o 7 2 o 2 cr 3 9. 10h 4zn no 3 4zn2 nh 4 3h 2o 3. Balance each of the following half cell reactions.
Balancing REDOX Reactions: Learn and Practice Reduction-Oxidation reactions (or REDOX reactions) occur when the chemical species involved in the reactions gain and lose electrons. Oxidation and reduction occur simultaneously in order to conserve charge. We can "see" these changes if we assign oxidation numbers to the reactants and products.
WS # 4 Balancing Redox Reactions . Balance each of the following half-cell reactions. (In each case assume that the reaction takes place in an ACIDIC solution.) Also, state whether the reaction is oxidation or reduction. 1.
Write balanced equations for the following redox reactions: a. 2 NaBr + Cl 2 2 NaCl + Br 2 b. Fe 2 O 3 + 3 CO 2 Fe + 3 CO 2 in acidic solution c. 5 CO + I 2 O 5 5 CO 2 + I 2 in basic solution ; Write balanced equations for the following reactions: a. Cr(OH) 3 + Br 2 CrO 4 2-+ Br-in basic solution 10 OH-+ 2 Cr(OH) 3 + 3 Br 2 2 CrO 4 2-+ 8 H 2 O ...
Questions pertaining to redox reactions. 1 in combination with nonmetals o n. Cr 2o 7 2 cr3 5. No no 3 6. Balancing redox reactions worksheet 1. Clo3 cl æ cl2 clo2. 1 in all compounds 2. A change in phase. H 2o 2 cr 2o 7 2 o 2 cr 3 9. Choose a method and complete questions 12 13.
Worksheet # 5 Balancing Redox Reactions in Acid and Basic Solution Balance each half reaction in basic solution. 4. Cr 2O 7 2 - → Cr3+ 5. NO → NO 3-6. SO 4 2- → SO 2 7. MnO 2 → Mn 2O 3 Balance each redox reaction in acid solution using the half reaction method. 8. H 2O 2 + Cr 2O 7 2- → O 2 + Cr 3+ 9. TeO 3 2-+ N 2O 4 → Te + NO 3-10 ...
! 211!! ThehalfJreaction!method!involves!balancing!the!oxidation!reaction!as!if!it! wereanisolatedreaction.Thenthereductionhalf Jreaction!isbalancedasifit!were
















![2-5 Redox Reactions Practice Worksheet With Answers [klzzqq3gwqlg]](https://idoc.pub/img/crop/300x300/9n0kgpw6zk4v.jpg)














0 Response to "38 redox reaction worksheet with answers"
Post a Comment